Pond Talk is an Amazon Associate. As an Amazon Associate we earn from qualifying purchases.
The silent killer hiding at the bottom of your pond. When the sun heats the surface, the bottom of your pond loses oxygen and turns toxic. Do you know how to break the seal before it’s too late?
Pond management often focuses on visible metrics such as surface algae or water clarity. However, the most critical physical process occurring in a deep water body is invisible to the naked eye. This process, known as thermal stratification, acts as a physical barrier that prevents the vertical exchange of gases and nutrients.
When a pond stratifies, it effectively splits into two separate ecosystems. The upper layer remains oxygenated through atmospheric contact and photosynthesis. The lower layer becomes an isolated, anaerobic chamber where organic matter decomposes into hazardous chemical byproducts. Understanding the mechanics of this “seal” is the first step in maintaining a stable, high-production aquatic environment.
## Pond Stratification Explained: Why Your Water Turns Toxic in Summer
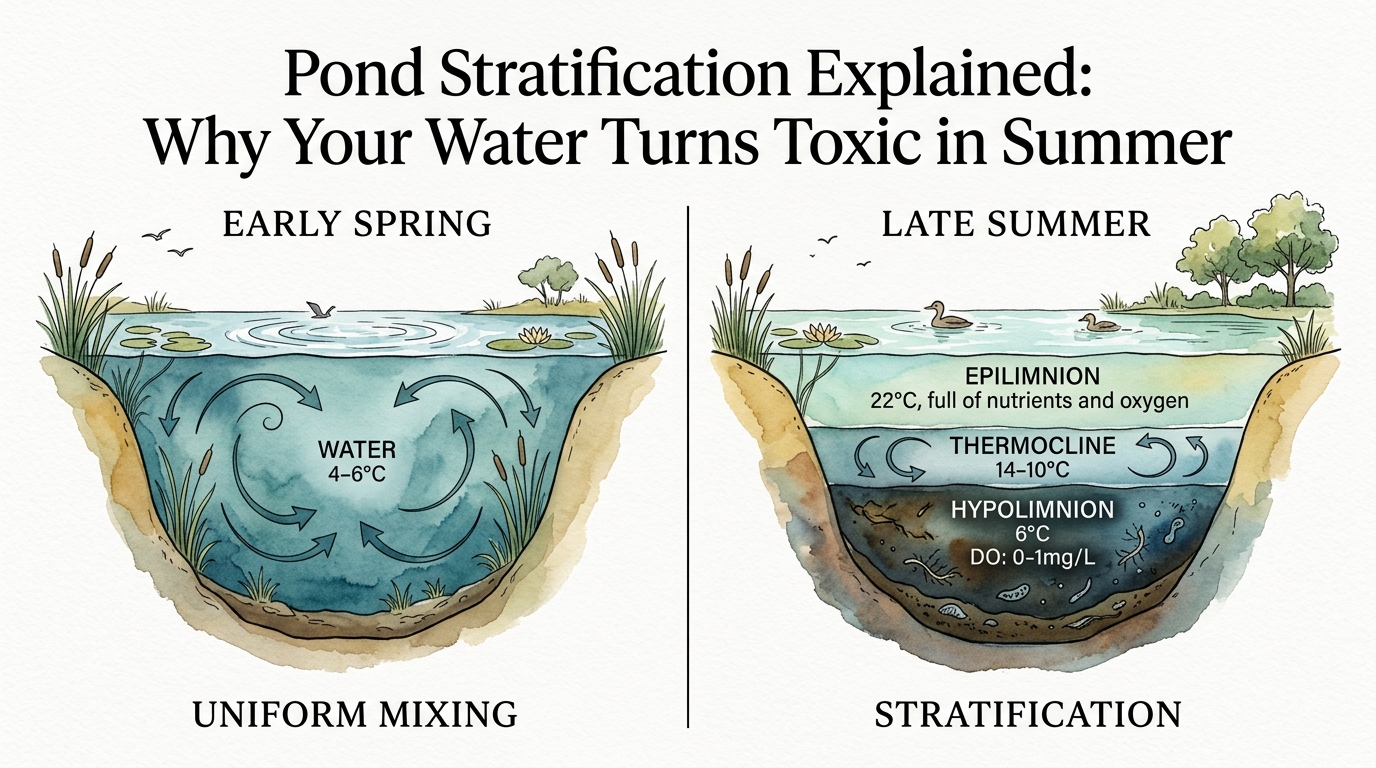
Pond stratification is a vertical layering of water caused by temperature-driven density differences. In freshwater systems, water reaches its maximum density at 3.98°C (approximately 39°F). As the sun warms the surface during the summer months, the top layer of water becomes less dense and floats upon the colder, heavier water at the bottom.
This density gradient creates three distinct thermal zones. The **epilimnion** is the top-most layer, which is warm, well-mixed by wind, and rich in dissolved oxygen (DO). Directly below this is the **metalimnion**, or thermocline, characterized by a rapid drop in temperature over a small vertical distance. The bottom layer, known as the **hypolimnion**, is cold, dense, and stagnant.
The metalimnion acts as a physical “seal” or membrane. Because the density difference is so great, wind energy at the surface is insufficient to push oxygenated water down into the hypolimnion. Without a source of fresh oxygen, the bottom layer begins to consume its own reserves. Bacteria decomposing organic matter—such as fish waste, dead algae, and leaf litter—rapidly deplete the remaining DO.
Once the hypolimnion reaches anoxia (zero oxygen), the chemistry of the pond shifts from aerobic to anaerobic decomposition. This shift triggers the production of toxic substances. In an aerobic environment, decomposition produces carbon dioxide (CO2). In an anaerobic environment, the process yields methane (CH4), ammonia (NH3), and hydrogen sulfide (H2S). Hydrogen sulfide is particularly lethal to aquatic life, even at concentrations as low as 0.5 to 1.0 parts per million (ppm).
## How Thermal Stratification and Aeration Work
The fundamental goal of mechanical aeration is to disrupt this stratification and facilitate a uniform water column. To achieve this, practitioners utilize the principles of gas transfer and mechanical mixing. The efficiency of this process is measured by the Standard Oxygen Transfer Rate (SOTR) and the Standard Aeration Efficiency (SAE).
SOTR represents the amount of oxygen an aerator adds to water in one hour under standard conditions (20°C, 1 atm, and 0 mg/L DO). SAE is calculated by dividing the SOTR by the power requirement (SOTR / Horsepower). These metrics allow for the comparison of different mechanical systems.
**Diffused Aeration Systems**
These systems utilize an on-shore compressor to pump air through a weighted line to a diffuser located at the pond bottom. As the bubbles rise, they create a “bubble plume.” This plume does not simply add oxygen through the bubbles themselves; instead, it utilizes the friction of the rising bubbles to pull the cold, deoxygenated water from the bottom to the surface. This mechanical lift breaks the thermocline and exposes the toxic bottom water to the atmosphere for gas exchange.
**Surface Aerators**
Surface aerators, such as paddlewheels or vertical pump sprayers, work by splashing water into the air. This increases the surface area available for oxygen absorption and facilitates the venting of gases like CO2 and H2S. While highly efficient at oxygenating the surface, they may lack the “reach” required to destratify ponds deeper than 10–12 feet unless they are specifically designed as high-volume circulators.
**The Physics of Oxygen Solubility**
Oxygen transfer is governed by Henry’s Law, which states that the amount of dissolved gas in a liquid is proportional to its partial pressure. At 0°C, pure water can hold approximately 14.6 mg/L of DO at saturation. At 30°C, this capacity drops to 7.5 mg/L. This inverse relationship means that during the peak of summer, when the water is hottest, the pond’s capacity to hold oxygen is at its lowest, while the metabolic demand of the fish is at its highest.
## Benefits of Mechanical Destratification
Implementing a system to break the thermal seal provides measurable improvements to the pond’s efficiency and health. By maintaining a uniform temperature and oxygen profile, the pond can support a higher biomass and resist sudden environmental shocks.
Uniform Habitat Availability: In a stratified pond, fish are often “squeezed” between the hot, high-oxygen surface and the cold, toxic bottom. Destratification allows fish to utilize the entire water column, reducing stress and improving growth rates.
Reduced Chemical Toxicity: Continuous mixing prevents the buildup of hydrogen sulfide and ammonia in the bottom sediments. This significantly reduces the risk of a “sudden turnover” fish kill, where a storm or cold front causes the layers to flip overnight, mixing toxic water into the entire pond.
Improved Nutrient Cycling: Aerobic bacteria are far more efficient at processing organic waste than anaerobic bacteria. By keeping the bottom of the pond oxygenated, the rate of “muck” decomposition increases, and the release of phosphorus from the sediments is inhibited. Phosphorus is the primary driver of nuisance algal blooms; keeping it locked in the sediment is a key strategy for long-term water quality.
## Challenges and Common Mistakes in Aeration
The most dangerous mistake a pond owner can make is starting a bottom-diffused aeration system in the middle of a hot summer without a proper “start-up” procedure. If the pond is already heavily stratified, turning on a powerful aerator will instantly pull the toxic, deoxygenated water to the surface.
This sudden mixing can cause an immediate oxygen crash throughout the entire water column, leading to a massive fish kill within minutes. To avoid this, a phased start-up is required.
- Day 1: Run the system for 30 minutes.
- Day 2: Run for 1 hour.
- Day 3: Run for 2 hours.
- Continue doubling the run-time until the water column is uniform.
Another common challenge is “under-sizing” the equipment. Aeration requirements are not based solely on the size of the pond but also on the Biological Oxygen Demand (BOD) and Sediment Oxygen Demand (SOD). A pond with a high organic load (lots of fish and muck) requires significantly more SOTR than a clean, sandy-bottom pond of the same volume. Using a system with insufficient SAE will lead to high energy costs without achieving the necessary destratification.
## Limitations of Pond Aeration Systems
Mechanical aeration is an effective tool, but it has physical and environmental limitations. In extremely deep water bodies (over 40–50 feet), the amount of energy required to achieve full destratification may exceed the economic benefit. In these cases, managers may opt for “hypolimnetic aeration,” which adds oxygen to the bottom layer without breaking the thermal seal.
Environmental limitations also include the risk of “thermal pollution.” In cold-water fisheries (such as trout ponds), breaking the stratification in summer can warm the entire water column to levels that are lethal for the fish. In these scenarios, the cold hypolimnion is a necessary refuge, and aeration must be handled via surface systems or pure oxygen injection that does not disrupt the thermocline.
Maintenance is a persistent limitation of diffused systems. Fine-bubble membranes are prone to “bio-fouling,” where algae and minerals clog the tiny pores. This increases the backpressure on the compressor, reducing SAE and potentially leading to mechanical failure. Regular acid cleaning or membrane replacement is a required operational cost.
## Comparison: Early Spring vs. Late Summer Profiles
The mechanical needs of a pond shift dramatically as the season progresses. The following table compares the typical physical and chemical profiles of a temperate pond.
| Parameter | Early Spring (Uniform) | Late Summer (Stratified) |
|---|---|---|
| Thermal Gradient | Minimal (< 2°C difference) | Extreme (> 10°C difference) |
| Oxygen Distribution | Uniformly high (8-10 mg/L) | Epilimnion: 6-8 mg/L | Hypolimnion: 0 mg/L |
| Toxin Accumulation | Zero to negligible | High concentrations of H2S and CH4 |
| Aeration Priority | Low (Natural wind mixing) | Critical (Mechanical lift required) |
| Mixing Resistance | Low (Isothermal state) | High (Thermocline barrier) |
## Practical Tips for Aeration Optimization
To maximize the efficiency of an aeration system, consider the following technical best practices:
- Diffuser Placement: Place diffusers in the deepest part of the pond. This maximizes the “rise time” of the bubbles, which increases the amount of water entrained and moved to the surface.
- Nocturnal Operation: In ponds with heavy algae blooms, oxygen levels are highest during the day (due to photosynthesis) and lowest at night (due to plant respiration). Running aerators exclusively from dusk to dawn can save energy while protecting fish during the critical DO “dip.”
- Monitoring ORP: Oxidation-Reduction Potential (ORP) is a better indicator of bottom health than DO alone. An ORP reading below -200 mV indicates highly anaerobic conditions, while a reading above +200 mV indicates healthy aerobic decomposition.
- Sizing for Peak Heat: Always size your aeration system for the hottest month of the year. Oxygen demand increases exponentially with temperature. A system that works in June may fail in August.
## Advanced Considerations: The Math of Oxygen Demand
Serious practitioners must account for the total oxygen budget of the pond. The total oxygen demand is the sum of fish respiration, phytoplankton respiration, and Sediment Oxygen Demand (SOD).
SOD is a critical variable. In a eutrophic pond, the sediment can consume between 1.0 and 8.0 grams of oxygen per square meter per day. If a pond has a surface area of 4,000 square meters (approximately 1 acre) and an SOD of 3.0 g/m²/day, the sediment alone consumes 12,000 grams (12 kg) of oxygen every 24 hours.
To counteract this, the aeration system must have an SOTR that exceeds this total demand, adjusted for site-specific factors (the “Alpha” and “Beta” factors). The Alpha factor represents the ratio of oxygen transfer in pond water versus clean water, typically ranging from 0.6 to 0.9. Failure to include these derating factors in your calculations will result in a system that is chronically underpowered.
## Scenario: The Summer Storm Flip
Consider a 1-acre, 15-foot deep farm pond that has been stratified for three months. The epilimnion (top 5 feet) is 85°F with 7.0 mg/L DO. The hypolimnion (bottom 10 feet) is 60°F with 0 mg/L DO and high levels of hydrogen sulfide.
A sudden thunderstorm arrives, dropping 3 inches of cold rain and bringing 40 mph winds. The cold rainwater is denser than the warm surface water and begins to sink rapidly. This physical displacement, combined with wind-driven waves, shatters the thermocline.
The 10 feet of toxic water mixes with the 5 feet of oxygenated water. Mathematically, the 7.0 mg/L DO in the top third of the pond is diluted by the two-thirds of the pond with 0 mg/L DO. The resulting average DO is 2.3 mg/L. However, the chemical oxygen demand (COD) of the accumulated toxins (H2S and CH4) immediately consumes another 1.5 mg/L. The final DO level drops to 0.8 mg/L. At this level, sensitive species like bass and bluegill suffer a 90% mortality rate within hours. This scenario is preventable through proactive, continuous mechanical destratification.
## Final Thoughts
Managing a pond effectively requires a shift from reactive observation to proactive mechanical control. Thermal stratification is a natural physical certainty in any sufficiently deep water body, but its consequences—anoxia and chemical toxicity—are manageable through engineered solutions.
By understanding the relationship between water density, oxygen solubility, and mechanical lift, you can maintain a pond that is healthy from the surface to the sediment. Investing in a high-efficiency aeration system with a strong SAE rating is not merely an aesthetic choice; it is a critical safeguard for the biological and chemical stability of your aquatic ecosystem.
Experiment with different diffuser placements and monitoring tools to find the optimal balance for your specific environment. The more data you collect regarding your pond’s thermal and oxygen profiles, the more precisely you can tune your systems for maximum efficiency. Proper management ensures that the “silent killer” at the bottom of your pond stays permanently at bay.
Amazon and the Amazon logo are trademarks of Amazon.com, Inc, or its affiliates.